Bonding (intra/inter)
|
What bonding (intra/inter) exists in the material?
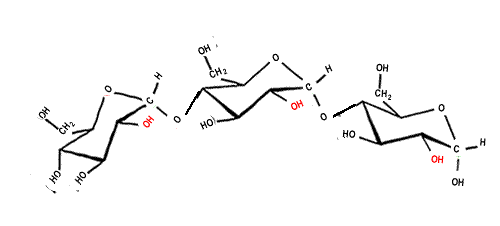
There are no specific intra or inter molecular bonds, however hydrogen bonding can exist as an intermolecular force between glucose molecules. This takes place with amylose, however most of the OH groups which are capable of forming hydrogen bonds project inwards, so only little hydrogen bonding can occur between the polymers of starch. This is one of the reasons why starch lacks structural properties compared to cellulose. The linkages between glucose molecules can also be considered as intra-molecular bonds. These bonds are known as glycosidic bonds, which are a type of covalent bond. Glycosidic bonds exist only in carbohydrates (sugars). These bonds can be formed by condensation (the removal of a water molecule), and broken by hydrolysis. |
|
|
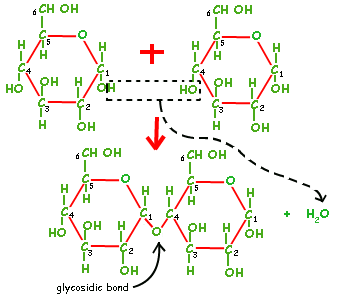
The image to the left is the bonding of alpha glucose molecules that are found in starch through condensation (1-4 glycosidic bond). This process occurs in the 1st and 4th carbon places of alpha glucose molecules, hence named 1-4 glycosidic bond. This bond occurs with the removal of a water molecule, also known as condensation.
|